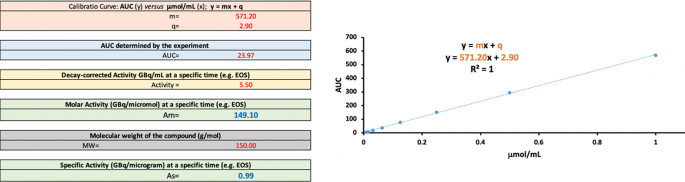
EANM guideline for harmonisation on molar activity or specific activity of radiopharmaceuticals: impact on safety and imaging quality | EJNMMI Radiopharmacy and Chemistry | Full Text

Calculating Radiation Exposures during Use of 14C-Labeled Nutrients, Food Components, and Biopharmaceuticals To Quantify Metabolic Behavior in Humans | Journal of Agricultural and Food Chemistry